
Dermorphin Peptide: Research in Mu-Opioid Receptor Agonism
May 12, 2026
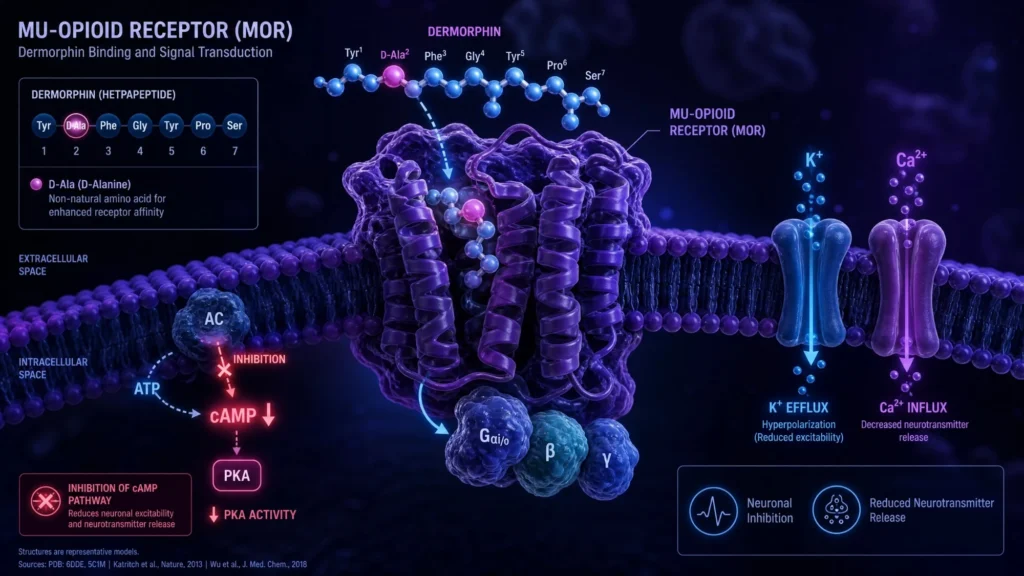
The Dermorphin peptide is a natural heptapeptide that research indicates is an exceptionally potent mu-opioid receptor (MOR) agonist. Originally isolated from the skin of South American tree frogs (Phyllomedusa sauvagei), Dermorphin is notable for being one of the first known peptides found in vertebrates to contain a D-amino acid, specifically D-alanine. Extensive preclinical studies have indicated that Dermorphin possesses analgesic potencies significantly higher—by some estimates 30 to 40 times higher—than morphine when administered centrally.(1) The precise molecular mechanisms underlying these effects are centered on its high affinity and selectivity for the $\mu$-type opioid receptor.
What is the mechanism by which Dermorphin exerts its effects?
The mechanism underlying Dermorphin’s proposed neuro-modulatory action appears to involve its highly selective interaction with mu-opioid receptors (MOR) throughout the central nervous system. Upon presentation, the Dermorphin peptide appears to bind to these receptors, potentially triggering the inhibition of adenylate cyclase. This interaction appears to cause a decrease in intracellular cAMP levels, the closure of voltage-gated calcium channels, and the opening of potassium channels. This cascade is considered crucial for hyperpolarizing the neuron and inhibiting the transmission of nociceptive (pain) signals.
Recent investigations suggest that the D-alanine at the second position of the peptide sequence is essential for its biological activity. This unique D-configuration appears to provide significant resistance to degradation by proteolytic enzymes, potentially allowing for a sustained interaction with the receptor compared to endogenous L-amino acid peptides.(1) (2)
How was the Dermorphin peptide discovered?
Professor Vittorio Erspamer and his research team at the University of Rome made significant strides in the late 1970s and early 1980s in elucidating the bioactive components of amphibian skin. They observed that certain tree frogs possessed skin secretions with intense pharmacological activity on the central nervous system.
In their investigations, Erspamer’s team isolated a novel substance which they named Dermorphin. Further analysis revealed that Dermorphin might exert a suppressive effect on pain perception by selectively targeting MOR. Building upon these findings, they characterized its unique sequence ($Tyr-D-Ala-Phe-Gly-Tyr-Pro-Ser-NH_2$), which appeared to represent a breakthrough in understanding how non-ribosomal-like modifications (D-amino acids) occur in vertebrate-derived peptides.(3)
Research Studies on Dermorphin Peptide
Dermorphin Peptide and Receptor Binding Affinity
The role of Dermorphin in opioid receptor binding was investigated to elucidate its potential for high-selectivity signaling. Using competitive binding assays in rodent models, the impact of Dermorphin was evaluated on receptor displacement and signal transduction. This study suggested that Dermorphin may exhibit a significantly higher affinity for $\mu$-receptors compared to $\delta$ (delta) or $\kappa$ (kappa) receptors. Histological analyses revealed that upon Dermorphin introduction, it may potentially result in a stabilized receptor-agonist complex, probably leading to more efficient G-protein activation. These findings suggest Dermorphin is a “highly specific tool for mapping mu-opioid receptor distribution and function.” (4)
Dermorphin Peptide and Neuro-metabolic Stability
This study aimed to investigate the role of D-alanine as a protective regulator against enzymatic degradation. Results indicate that Dermorphin potentially interacts with neural enzymes differently than standard L-peptides, resulting in potential negative regulation of degradation pathways. In vitro experiments using neuronal cultures suggest that the peptide may play a role in sustained inhibitory signaling, as indicated by potential differential regulation of neurotransmitter release. Research further suggests that by disrupting the standard metabolic clearance of opioid peptides, Dermorphin appears to activate the MOR pathway for longer durations, potentially providing a unique model for studying chronic receptor activation and desensitization in mice.(5)
Synopsis
The Dermorphin peptide appears to be a promising modulator of the mu-opioid signaling system. By utilizing its unique D-amino acid configuration to resist enzymatic breakdown, Dermorphin appears to activate the MOR pathway with extreme potency, potentially leading to enhanced antinociceptive responses in research models. Studies have suggested its potential in mapping receptor populations and exploring the fundamental mechanisms of opioid signaling. Further investigations are warranted to fully elucidate its underlying mechanisms and evaluate its applications in scientific research.
NOTE: These products are intended for laboratory research use only. This peptide is not intended for personal use. Please review and adhere to our Terms and Conditions before ordering.



